Cocaine Break Down
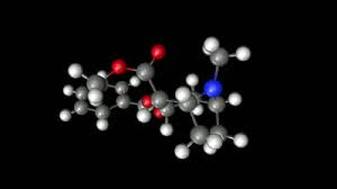
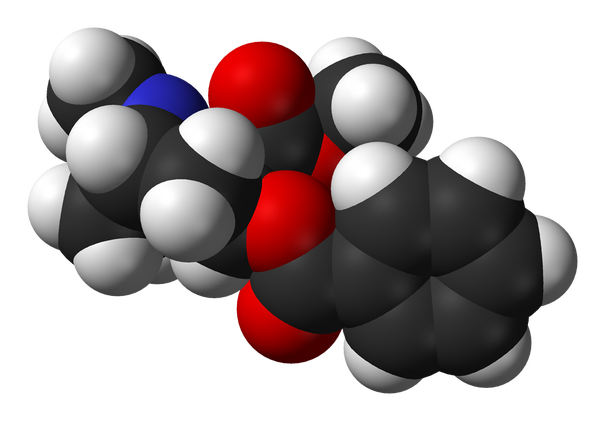
The different elements that make up cocaine are carbon, hydrogen, nitrogen and oxygen. There are 17 carbons, 21 hydrogens, 1 nitrogen and 4 oxygens. Cocaine is made up of all non-metals, even though hydrogen is on the opposite side of the periodic table it is still considered a non-metal.
Cocaine is divided into two sub groups which are named cocaine hydrochloride and cocaine free base. Cocaine hydrochloride's physical properties include the molecule being colorless or white, it is odorless and bitter tasting in solid form. In water its solubility is 200 grams per 100 mls At room temperature cocaine hydrochloride is solid-crystal form and has a melting point of 197 degrees celsius. Cocaine freebase is white and just like cocaine hydrochloride it is a solid-crystal in its natural state. The molecule is slightly volatile, anhydrous, and also has a bitter taste. Cocaine freebase's solubility in water is .17 g per 100 ml's, it also has a melting point of 98 degrees celsius and a boiling point of 188 degrees celsius. The solution that cocaine is found in is the cocaine hydrochloride which can be used as an anesthetic because it blocks the initiation or conduction of nerve impulses. This solution can be absorbed from all sites on the body including mucous membranes of the oral laryngeal and nasal cavities. Although cocaine is slightly soluble is water it dissolves rapidly in alcohol, ether, or chloroform.
Street Names etc.
Cocaine has acquired a numerous amount of street names, and it is based on the way that the cocaine is used. Some names include badrock which refers to cocaine powder, cakes which is smokeable crack/cocaine, happy powder, moon rocks, nose candy, snow, stardust, and zip which is injected cocaine powder. Also the ones most people have heard of which would be crack cocaine.
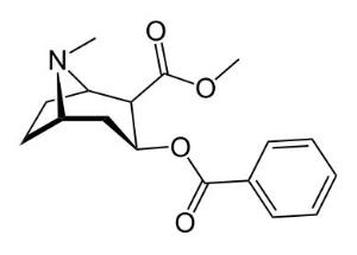
Cocaine is an organic molecule based on the fact that it has 17 carbons. Organic groups that are in cocaine are 2 esters, benzene and amines.
The molecular formula of Cocaine is C17H21NO4. The molecular weight is 303.53 grams per mole. The percent composition of cocaine is 67.30% carbon, 6.97% hydrogen, 21.09% oxygen and 4.62% nitrogen.
"Cocaine Hydrochloride Topical Solution - FDA Prescribing Information, Side Effects and Uses." Cocaine Hydrochloride Topical Solution - FDA Prescribing Information, Side Effects and Uses. Drugs.com, 2000-2015. Web. 17 Sept. 2015.
"Cocaine." (PIM 139). International Program on Chemical Safety, n.d. Web. 17 Sept. 2015.
"Cocaine." Chemistry. Edinburgh: E. & S. Livingstone, 1919. 816-17. Print.
Heath. "Circle and Identify Functional Groups (Cocaine)." YouTube. YouTube, 3 Mar. 2012. Web. 17 Sept. 2015.
National Center for Biotechnology Information. PubChem Compound Database; CID=5760, https://pubchem.ncbi.nlm.nih.gov/compound/5760 (accessed Sept. 17, 2015).
Perkin, F. Mollwo. Qualitative Chemical Analysis, Organic and Inorganic. London: Longmans, Green, 1910. Print.
"Street Names." - In The Know Zone. Education Specialty Publishing, 2011. Web. 17 Sept. 2015.
"Cocaine." (PIM 139). International Program on Chemical Safety, n.d. Web. 17 Sept. 2015.
"Cocaine." Chemistry. Edinburgh: E. & S. Livingstone, 1919. 816-17. Print.
Heath. "Circle and Identify Functional Groups (Cocaine)." YouTube. YouTube, 3 Mar. 2012. Web. 17 Sept. 2015.
National Center for Biotechnology Information. PubChem Compound Database; CID=5760, https://pubchem.ncbi.nlm.nih.gov/compound/5760 (accessed Sept. 17, 2015).
Perkin, F. Mollwo. Qualitative Chemical Analysis, Organic and Inorganic. London: Longmans, Green, 1910. Print.
"Street Names." - In The Know Zone. Education Specialty Publishing, 2011. Web. 17 Sept. 2015.